Zinc Earthing Cell
The importance of control of stray current is becoming more apparent as the pipeline network is globally expanded. Sources from which these stray currents emanate can be put down to a variety of reasons, from both an AC and DC basis. Super grid electricity transmission system is of particular concern. Increasingly zinc grounding cells are used as earthing material on sights where corrosion has occurred or ‘is’ induced.
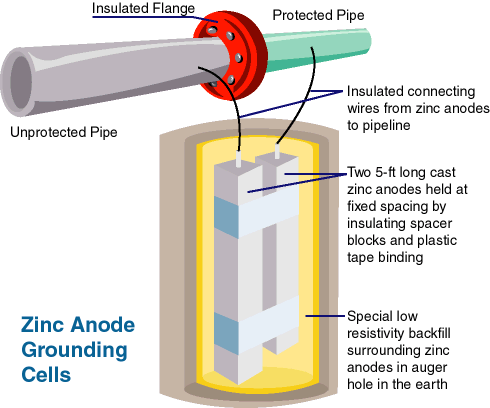
Zinc grounding cells provide a low resistance across an insulating joint without introducing excessive loss of cathodic protection current by developing a polarization potential across the cell of several tenths of a volt that has the effect of greatly reducing flow of cathodic protection current through the cell.
These cells consist of two zinc rods, with insulation material to separate them, in a conductive backfill material, packaged in a bag. The insulation or separator leaves the zinc rods reasonably close together without touching. The backfill material, such as bentonite, promotes conduction and contact with the surrounding earth, as the package is buried near the points it is connected to.
Chemical composition of Zinc Electrode:
Copper Cu 0.005% max
Aluminum Al 0.10 – ۰٫۵۰%
Iron Fe 0.005% max
Cadmium Cd 0.025% – 0.07%
Lead Pb 0.006% max
Others (total) 0.10% max
Zinc Zn Remainder
Potential Ag/AgCl – 1.05 V
Capacity ampere hours 780 per kg min
Packaged in cotton bag with the following chemical backfill Composition:
Powdered Gypsum 75%
Granulated Bentonite 20%
Sodium Sulphate 5%electrical efficiency and long working life
A wire lead attaches each rod to a point of interest, such as across an insulated flange, or between a pipeline and ground. The concept for protection is that the zinc rods will allow separation of the two structures, and minimize the DC cathodic protection current flow under normal conditions. Under over-voltage conditions, the small separation between the rods and the conductive backfill . will allow current to flow between them, draining the un
